|
Maggots, although often seen as disgusting, constantly wriggling little creatures, are as alive as we are, and, like us, also exhale carbon dioxide. Thus, our class created a little experiment involving these larvae, to measure the amount of CO2 that they exhale, depending on a certain variable. My group has chose to use light as the independent variable. Thus, our experiment went like this: First, with a pipette, we poured 2.5 mL of hydrogen carbonate indicator at the bottom of two respiration chambers. We also laid five maggots on the little platform of the chamber, then we closed everything. One respiration chamber was put under the light of a lamp, while the other chamber was put in a space where there was less light. We then waited for twenty minutes for something to happen. And something did happen! On the photo below, you will see that the respiration chamber on the left, which had not been under the lamp, contains the hydrogen carbonate indicator that has not changed colour since the beginning of the experiment. However, the indicator of the chamber on the right did change colour; it turned slightly orange. According to the table of the hydrogen carbonate indicator, this means that the pH level is at 8.2, instead of the normal pH of 8.4. This is not a big change in pH, however it does mean that there is an increase in CO2.
Thanks to this experiment, we can conclude that the maggots that were under the lamp exhaled more CO2, than those in the dark. However, there is one variable that my group did not think of: of course, putting maggots under the light of a lamp with no protection means that the maggots will also be under more heat. Therefore, the result could also mean that heat increases the rate of CO2. Another variable that had to be thought of is that of some maggots probably having died, even before we started the experiment. This would also have altered our results, because the fewer the maggots are breathing, the least CO2 there is inside the chamber. However, we can still conclude that the maggots do breathe out CO2, and that light and heat accentuate the rate of this process. if you are interested in knowing more about this, some of the other groups in my class have also experimented this with different variables, such as stressful and non-stressful music, or with extremes of temperature. Below are a couple photos of my experiment, enjoy the view of these wonderful maggots!
0 Comments
After the discovery of the structure of DNA, Meselson and Stahl found evidence for semi-conservative replication in DNA.
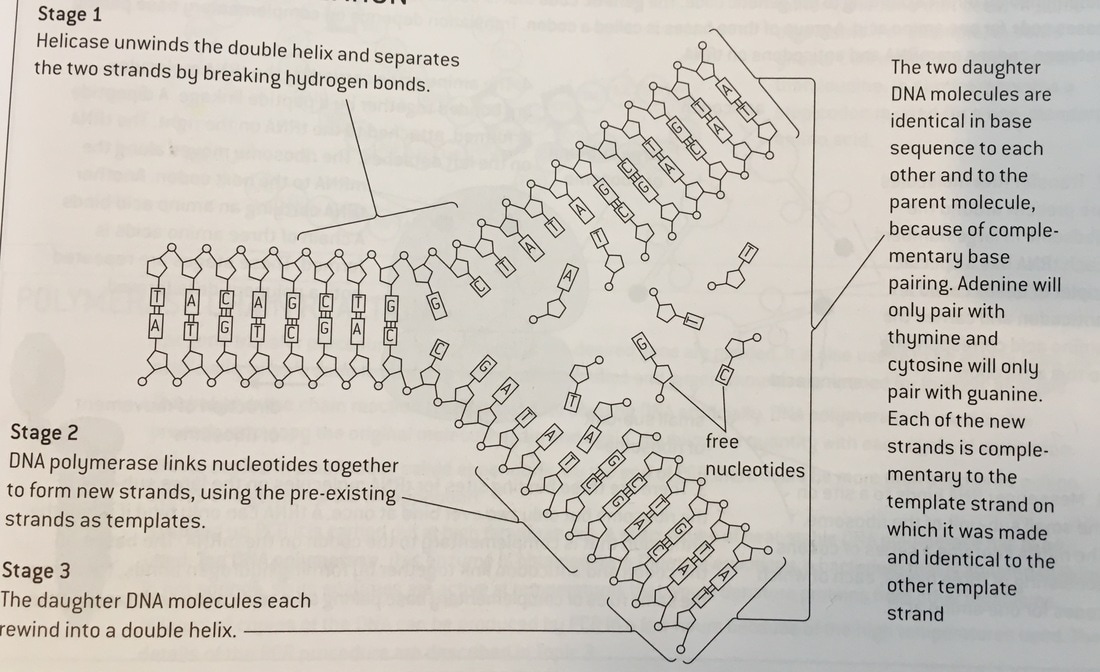
They found this out by culturing E. coli bacteria in a medium where the only nitrogen source was Nitrogen 15 for many generations. The nitrogen in the bases of the bacterial DNA was also Nitrogen 15. They then abruptly transferred the bacteria to a medium with the Nitrogen 14 isotope, which is less dense. They spun a solution of caesium chloride in an ultracentrifuge at 45,000 revolutions per minute for 24 hours. Because the caesium ions are heavy, they tend to sink, therefore establishing a gradient with the greatest caesium concentration, with the density at the bottom. Meselson and Stahl then spun samples of the DNA collected from their bacterial culture at different times after the transfer to the Nitrogen 14 medium. They could examine the DNA because it shows up as dark in UV light. Meselson and Stahl noticed that after one generation, the DNA had a density between Nitrogen 14 and Nitrogen 15, due to semi-conservative replication, which mixed up the DNA with one old and one new strand. After two generations, there were four bands, two with a density between N14 and N15, and two with a density of only N14. The following generations had more and more DNA bands with only N14, and fewer and fewer N15/N14 bands. This is all due to semi-conservative replication. This consists of the two strands of the DNA molecule being separated by the breaking of the hydrogen molecules between their bases, thanks to helicase. New polymers of nucleotides are assembled on each of the two single strands. A strand of DNA on which a new strand is assembled is called a template strand. Because of complementary base pairing, each of the new strands has the same base sequence as the old strand that had been separated from the template strand. This produces two DNA molecules that are identical to each other and to the original parent DNA molecule. Therefore it is called semi-conservative replication, because each of the DNA molecules produced has one new strand and one strand conserved from the parent molecule. Introduction To speed up chemical reactions, enzymes, which are biological catalysts, are used. They are made up of proteins and they are very efficient, thus being very useful in all processes in living organisms. Those include digestion, respiration and photosynthesis. They use the rule of the lock and key model, in which the active site on the enzyme molecule forms a sort of keyhole in which a substrate can fit in. Temperature and pH are very important for enzyme activity, but this experiment especially looks at temperature. If a temperature is too low, the reaction would not occur because there is not enough kinetic energy for the enzymes to collide and react, however if there is too much heat, the enzymes will denature, which means that the lock and key technique would not work anymore, because the active site has changed shape and the substrate cannot fit in anymore. Therefore, this investigation looks at the effect of one of these two factors, temperature, on the enzyme amylase. This is an enzyme found in the salivary glands. It is used to break down starch into maltose as part of digestion. To measure the activity, we see how long it takes for the starch to disappear, by testing for it with iodine. Aim The aim of this experiment is to find out the rate of reaction between amylase and starch in various temperatures. Hypothesis Because the reaction of amylase and starch is dependent on the temperature of the environment, my hypothesis is that as the temperature rises to its optimum level, the rate of reaction will increase. Once the temperature will rise above the optimum level, the rate of the reaction will decrease. This can be backed up by the fact that the temperature raises kinetic energy, and therefore the molecules are able to move around faster and would bump into each other, or collide, and react more easily. This is to follow the rule of the lock and key model. However, this is only possible if the temperature is at its optimum level due to the kinetic energy, and if the temperature is too high, the enzymes will be denatured. This means that the structural shape of the enzyme will be shaped, thus not enabling the lock and key process. Method For this investigation, a few apparatus and materials should be needed:
We used five different temperatures: 0C°; 20C°; 30C°; 35C°; 63C°. Independent variable: the temperature Dependent variable: the time Control variable: Iodine solution For each different temperature, we put 2mL of amylase with the aid of a pipette in one test tube, and 2mL of starch (we used our own saliva) in another test tube. We let both test tubes stay five minutes in the temperature that we were testing at. We then continued by mixing the two solutions together into one of the two test tubes, and let it stay five minutes in the temperature again. While we were waiting, we filled a dimple tray with a drop of iodine solution in each compartment. After five minutes, we put one drop of the starch and amylase solution in a different compartment every 30 seconds. We did this until there was no more solution to take from, or until we had filled up all the compartments. We counted the amount of time the solution took to change colour. If the solution changed colour, that means that the starch had disappeared. We then noted down the amount of time and continued. Risk assessments For this investigation, there are a few risk assessments that must be taken into account.
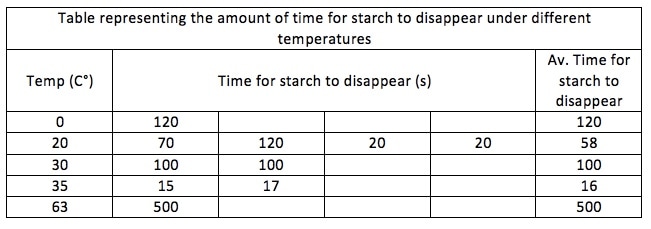
Results Here is a table that represents the time and average time for starch to disappear under different heating conditions: The table shows us that the optimum temperature for starch to disappear is 35C°, because it takes the least time (16 seconds). The table also shows that it takes increasingly less time for the starch to disappear as the temperature rises, and once the temperature goes past 35C° it takes increasingly more time. Not all the values in the table are entered, therefore it shows that it takes only 58 seconds for the starch to disappear at 20C°, where it takes 100 seconds at 30C°. The mean time for 20C° is higher due to the fact that there are more values entered than at 30C°.
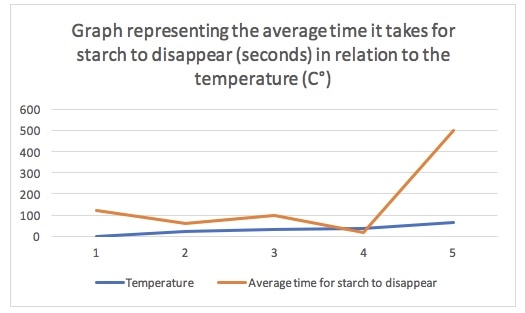
The graph has the same issue because it takes in the values for the temperature and for the average time for starch to disappear. Where the blue and the orange line are closest to each other is where starch takes the least time to disappear. The graph shows us that starch takes less and less time to disappear, and that there is therefore increasingly more activity, until the temperature rises too high for the enzymes to still react. Evaluation Since the optimum temperature for an enzyme to work in is 37C°, the results of this experiment are approximately right, because the enzyme worked best in 35C°. However, there have been gaps in the experiment, because some values for the amount of time it took for starch to disappear at a certain temperature were not entered, because of a lack of time or because there was not enough amylase and starch solution left. Therefore, the results for the average amount of time were not entirely accurate. This is best shown when it took more time for starch to disappear at a temperature of 30C°, than at a temperature of 20C°. This is supposed to be wrong, because the amount of time should decrease as long as the temperature increases towards its optimum level. The process was not entirely reliable either, due to certain limitations. These include the fact that the quantities of the different solutions were not measured exactly, and therefore some solutions contained more starch, amylase or iodine than others, which would alter the results and would produce less reliable data. Furthermore, the equipment was not accurate either: for example, the pipette did not have enough accuracy because of air bubbles often forming inside it, therefore making the measurement inaccurate, which would result in the same issue as earlier. There is also always room for human error: since we had to time thirty seconds until we would place a drop of the amylase and starch solution in the iodine solution, it would never be exactly thirty seconds, because humans have a slow reaction time. Human error was also possible in the area of senses and observation, because we could not see the colour change that would show there is a reaction at the exact moment it happened. Therefore, the timing was not precise enough, also resulting in imprecise results and data. Furthermore, the temperatures that the solutions were put in could never be exact and could also never stay constant, because the bodies that surrounded them would give off heat, and the air flow in the room would also shift them around. This would influence the experiment in the way that we would never be sure that, for example, 35C° is the exact optimum temperature for the enzyme to react. Finally, the processing of data can also often lead to anomalies that would influence processing in the graph. Therefore, the graph would not be entirely accurate, and the results of the experiment would not be 100% reliable. Therefore, it would not be an experiment that we could entirely apply to real life. To rectify these limitations in a similar experiment in the future, I would add more trials and data points to obtain more accurate and reliable results. I would also use more temperatures to test on, which also has to do with accuracy and reliability. Finally, the equipment should be digital, rather than analog, to have more precise measurements and results. In this experiment, our class immobilised the lactase in milk, to create a milk that cats would be able to digest.
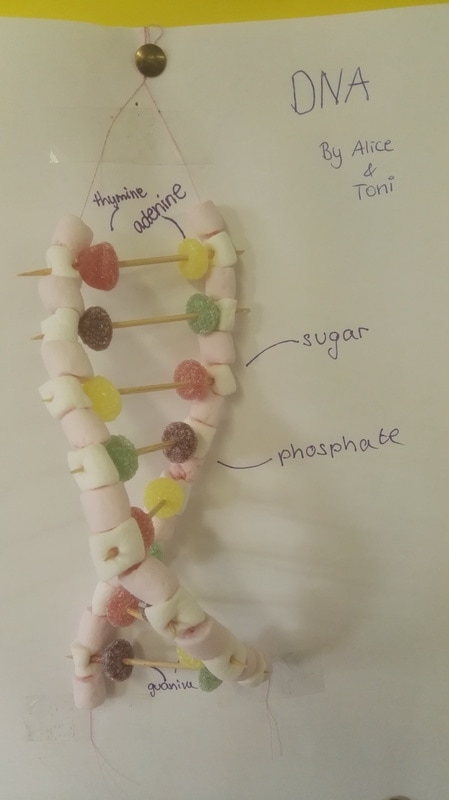
In the drawn representation of the monomer above, you can see how one monomer is formed, and look how they get linked to another one to form the antiparallel structure of DNA:
Water is one of the most important elements on Earth. This is because it has a large number of properties that make it very useful to living organisms, so much so that life could not exist without it. Here is a list of its properties:
Water is also important in blood transport. The mode of transportation of a substance depends on its solubility in water.
Water has a lot of properties, and these are the reasons why water is more useful than methane, which has a similar molecular mass, but has weaker intermolecular forces. The properties of methane are simply less useful than those of water:
To conclude, water has a lot of properties that help living organisms in many ways, due to its cohesiveness, adhesiveness, a high melting, boiling, latent heat vaporisation and specific heat capacity point, and it being a solvent. To look more specifically into examples of how it is used in living organisms, see the first paragraph where its properties are listed with specific examples. Here are a few links to some videos that might be interesting if you would like to learn more about water: Life is essentially made up of a complex web of chemical reactions. The discovery of DNA in the 1950s helped molecular biology on a very large scale. Living organisms essentially use hydrogen, carbon, nitrogen, oxygen, sulfur, phosphate and helium to create molecules, which are mainly carbohydrates, lipids, proteins and nucleic acids. Each atom in a molecule has a set number of links.
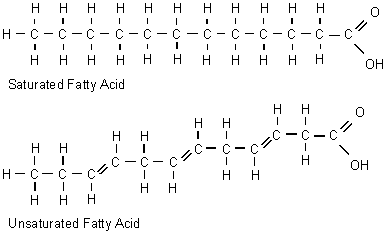
Following glucose, is ribose. Ribose is also a carbohydrate, therefore it also contains carbon, hydrogen and oxygen, only they are differently placed. A saturated fatty acid is a lipid, it can then have a double bond with oxygen.
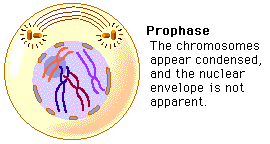
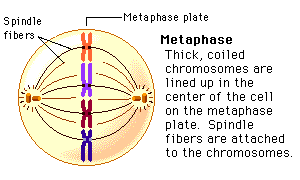
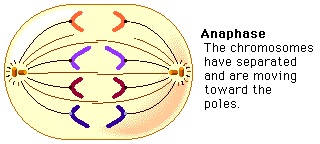
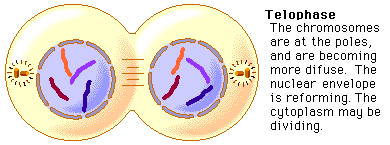
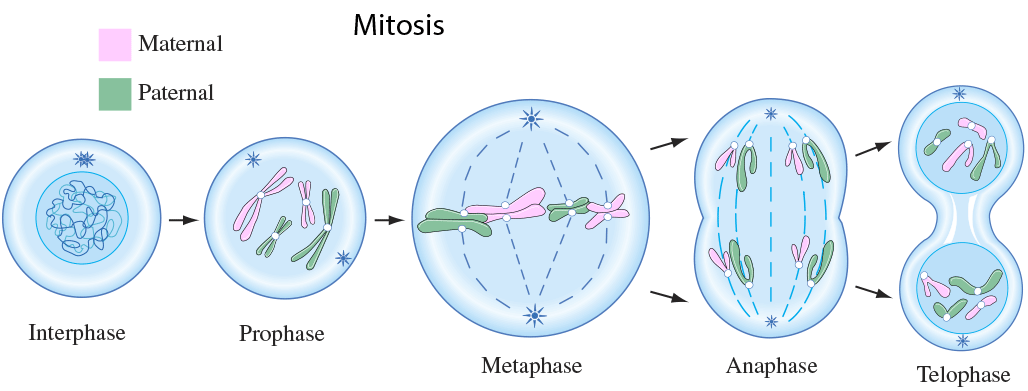
Amino acids are part of the protein group. They have a "R" group in the molecule, which varies from molecule to molecule. These were the main molecules found in the units of life. They can react with each other and have hugely varied roles within and outside of cells. It is important to know these four molecules for the exam, and to know how to draw them. And because carbon is a very important atom in each and every one of these molecules, here is a video to quickly learn more about the well-known atom. Mitosis is a type of cell division in which two new cells form through the separation of the chromosomes initially in the parent cell and the formation of to daughter cells in which the separated chromosomes are now situated. These daughter cells then have the same number and kind of chromosomes as in the parent nucleus. Mitosis is very important because it leads to growth or repair. To identify the way mitosis worked, our class decided to also have a little fun by drawing the different stages of mitosis on donuts with icing. Of course we ate some by the end of our drawing session. There are four stages of mitosis: prophase, metaphase, anaphase and telophase. The first phase is prophase, in which the chromosomes in the nucleus of the cell become shorter and fatter by supercoiling and the spindle microtubules in the cell also grow. In this phase the chromosomes are even individually visible. Here is a drawing of the prophase stage, as well as its equivalent on a donut: If we went a little more into details, we could find that there there are two phases in prophase itself: early and late prophase. Early prophase consists of the supercoiling of the chromosomes and the growth of the spindle microtubules, or also called the spindle fibres. In late prophase, the chromosomes become attached at the centre, called the centromere. Each chromosome consists of two identical chromatids. the spindle fibres also start extending from each pole of the cell towards the equator. The drawing of the cell above would show the chromosomes in late prophase. Second comes metaphase, in which the nuclear membrane containing the chromosomes is broken down and all the chromosomes have moved to the equator of the cell, where they align. The spindle microtubules then attach to each centromere of the chromosomes at the equator. The chromosomes would at this point still be called chromatids. Here is again a drawing of metaphase next to the edible drawing: After metaphase, the chromosomes are separated and pulled apart at the centromere by the spindle fibres in anaphase. The chromatids have now become chromosomes. Finally, in telophase, the chromosomes are all at either end of the pole of the cell and a nuclear membrane forms around them, after the spindle fibres have broken down. In this phase the cell also pinches inward to separate into two identical cells, each with an identical nucleus. The division of the cell is called cytokinesis. Inside the nucleus, the chromosomes uncoil and are no longer individually visible. Telophase is also a phase in which there is early and late telophase. The early phase is the one in which the two nuclei form inside the still whole cell, and late telophase consists of the pinching in of the cell wall to form two identical cells. Here are the two drawings: The image on the left is showing the process of early telophase, with the cell already visibly pinching in and slowly separating its two poles at the equator. On the right, the two cells are visibly separated. Cytokinesis is a process that varies depending on plant cells or animal cells. We have already looked at cytokinesis in animal cells, however plant cells go through this phase in a different way. In plant cells, a new cell wall is formed across the equator of the cell, with plasma membrane on both sides. This divides the cell in two. Below is a representation of this process: One can also measure the mitotic index in a tissue. This is the ratio between the number of cells in mitosis and the total number of cells in a tissue. To calculate this, one has to divide the number of cells in mitosis by the total number of cells in a tissue. This index is often used by doctors to detect tumours inside the body, how rapidly it will grow and therefore was treatment would be needed. To doctors, a high index would show a rapidly-growing tumour.
To conclude, here is the full process of mitosis as drawn normally and in a slightly different way - on doughnuts, the picture being taken with hands sticky from the icing and a grumbling stomach. An article published by the Daily Mail demonstrates why shaving might raise breast cancer risk.
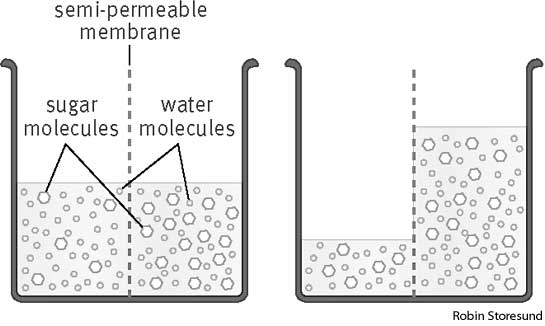
The article linked it to the use of anti-perspirant deodorant, and suggested that shaved skin might more easily absorb the cancerous chemicals. This was discovered through a study that examined 437 female breast cancer patients in the U.S. The women who used anti-perspirant or deodorant sprays or roll-ons as well as shaved, regardless of the number of times a week they did so, were on average 59 years old when a tumour was detected in their breast. In contrast, those who did not shave or use deodorant or anti-perspirant were on average not diagnosed with breast cancer until 67 years of age. The lead researcher suggested then that underarm shaving might play a role in breast cancer. This study is valid and reliable because it took in account a large number of women that have breast cancer, with one group that had shaved and used deodorants and a control group that had not shaved neither used deodorant. It is also a precise research because it described how it was carried out with a precise number of subjects. However, it does not explain thoroughly why shaving really increases breast cancer risk, it only vaguely explained that the pores are more open to chemicals, that may be aluminium compounds, to enter and cause breast cancer. Here is the link to the article: http://www.dailymail.co.uk/health/article-206459/Shaving-raise-cancer-risk.html Biology practical: osmosis as seen through hypertonic and hypotonic solutions Research Question: Osmosis is the passive movement of water molecules from a region of lower solute concentration to a region of higher solute concentration, across a partially permeable membrane, until an equal amount of water molecules is achieved on either side. This can be observed through a practical involving potatoes cut into pieces and soaked in solutions with different levels of sucrose in them. This practical’s aim was to study the effects of osmosis on these bits of potato and to find which solutions were hypotonic or hypertonic. Here is an example for osmosis: Preparation: The practical was carried out by cutting up three pieces of potato in roughly the same size, their length and mass being the independent variable. These potato bits were blotted with a tissue paper to take the excess water out of them, then were measured with a ruler and weighed with a balance and the information was written down in a table.
Results and analysis:
By the end of the practical, when the potato bits were taken out of the liquid, the length and mass had changed. Here are the new results, with the difference between the old and the new length and mass:
As one can see, the length of the potato bits had not changed.
However, the mass changed: Potato soaked in pure water: mass increased by 0.8g Potato soaked in mixed Ribena and water: decreased by 0.13g Potato soaked in pure Ribena: decreased by 0.52g Evaluation: This practical was carried out with as much precision as possible, however the amount of liquid in the test tubes was not exactly the same between them and this could have affected the results. The amount of sugary liquid mixed with the water in the mixed solution had not been calculated, and this could have affected the results as well, because the mixed solution could have contained either more water than Ribena or the opposite, which could consequently have made the solution either hypertonic or hypotonic compared to the potato piece. The time taken to let the potato bits soak in the water was also unmeasured and was not taken into account in the practical. This fails to show how much time it takes for a potato to grow or shrink size; to see what amount of liquid passes through the partially permeable membrane in a certain amount of time. But apart from a few imprecisions, the practical was carried out with no particular incidents and the results made sense. Conclusion: The water is hypotonic compared to the potato bit, which is then hypertonic. This means that more water molecules move in through the partially permeable membrane of the potato from the region of lower solute concentration, or equally called the region of high water potential (the water) to the region of higher solute concentration (the potato). Meanwhile, the solute concentration in the mixed and pure Ribena is higher than the one inside the potato chip, therefore the water molecules move from a region of lower solute concentration to one of higher solute concentration, which explains the decrease in mass in the potato bit. This means that this time the solution was hypertonic compared to the hypotonic potato bit. |
PaulineIB Biology student |